release time:2021-09-08 11:50:34

2021-12-29
Veterinary biochemical testing is the testing of multiple chemical components within a sample. The levels of these chemicals reflect many conditions in various organs of the body. Most veterinary biochemical tests measure blood electrolytes and assist in the diagnosis of liver, kidney and pancreatic disease.

2021-12-08
Dry chemistry analytical techniques are relative to wet chemistry techniques. Dry chemistry is the direct addition of a liquid test sample to a dry reagent produced specifically for a different project. The moisture of the sample under test is used as a solvent to cause a specific chemical reaction, which leads to a chemical analysis method. Dry chemistry is a class of analytical methods based on enzymatic methods. Dry chemistry can also be called solid phase chemistry. Dry chemistry uses reflectance photometry or differential electrode method as a measurement method.
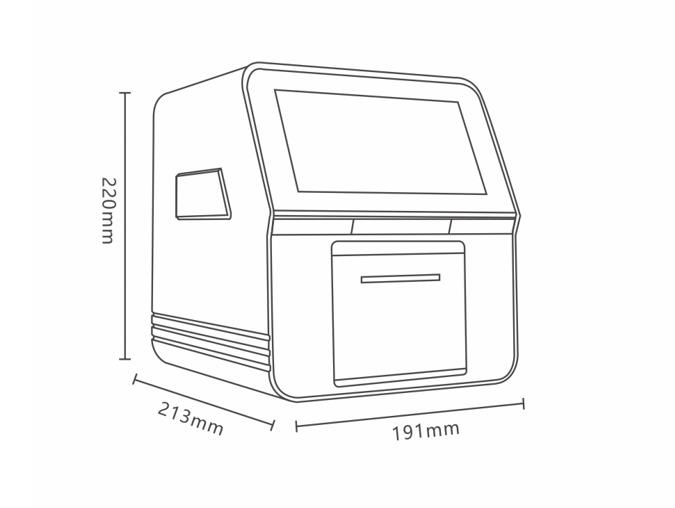
2021-08-12
The SMT-120 uses a real-time quality control management system, allowing doctors to have more accurate test data. The SMT-120 analyzer can be operated without the need for a centrifuge, manual addition of diluent, or a medical specialist.